ERP for Nutraceutical Manufacturers Under 21 CFR Part 111: What Compliance Really Requires

ERP for Nutraceutical Manufacturers Under 21 CFR Part 111: What Compliance Really Requires
TL;DR 21 CFR Part 111 holds dietary supplement and nutraceutical manufacturers responsible for proving the identity, purity, strength, and composition of every product they ship and the FDA cited a 46% increase in Part 111 observations between 2023 and 2024. Spreadsheets, paper batch records, and bolt-on quality tools cannot keep pace with cGMP expectations. inecta Food ERP gives nutraceutical manufacturers a single platform for batch production records, identity testing, potency and overage tracking, lot traceability, and automated COA generation all built into the system, not customized in.
Part 111 Is Not Optional, And Auditors Are Paying Closer Attention
If your company name appears on a supplement label, 21 CFR Part 111 applies to you. That includes own-brand manufacturers, contract manufacturers, private labelers, packagers, and holders. Outsourcing production does not transfer the responsibility. Neither does relying on a supplier's Certificate of Analysis as your sole identity check the FDA has been clear that a CoA alone is insufficient unless the supplier is qualified, the testing methods are documented, and quality control personnel review and approve the documentation.
The recurring pattern in FDA 483 observations is striking. Year after year, the most common Part 111 citations involve missing or inadequate component and finished product specifications, deficient master manufacturing records, and incomplete batch production records. These are not exotic compliance failures. They are documentation failures and documentation failures are exactly what an integrated ERP is designed to prevent.
What 21 CFR Part 111 Actually Demands From Your Systems
Part 111 spans 16 subparts covering personnel, physical plant, equipment, production and process controls, quality control, components, master manufacturing records, batch production records, laboratory operations, manufacturing operations, packaging, holding, returned product, and complaints. From a software standpoint, the core obligations come down to a handful of things your business systems must produce and preserve on demand:
- Specifications for the identity, purity, strength, and composition of every component and every finished batch, including limits on potential contaminants.
- Identity verification of every dietary ingredient at receiving, with documented test methods and results.
- Master Manufacturing Records (MMRs) that define exactly how each product is made.
- Batch Production Records (BPRs) that prove each batch was manufactured according to the MMR, with all in-process checks, deviations, and dispositions captured.
- Quality control approvals at every disposition point — receiving, in-process, finished product release, returned product, and complaint investigation.
- Lot traceability forward and backward across the supply chain, sufficient to support a recall.
- Records retention under Subpart P, with full reviewability during inspection.
Each of these is a data problem. And data problems get worse, not better, when the answers live in disconnected spreadsheets, email threads, paper folders, and standalone QC software that does not talk to your inventory or finance system.
Why Generic ERP Is Not Enough For Nutraceuticals
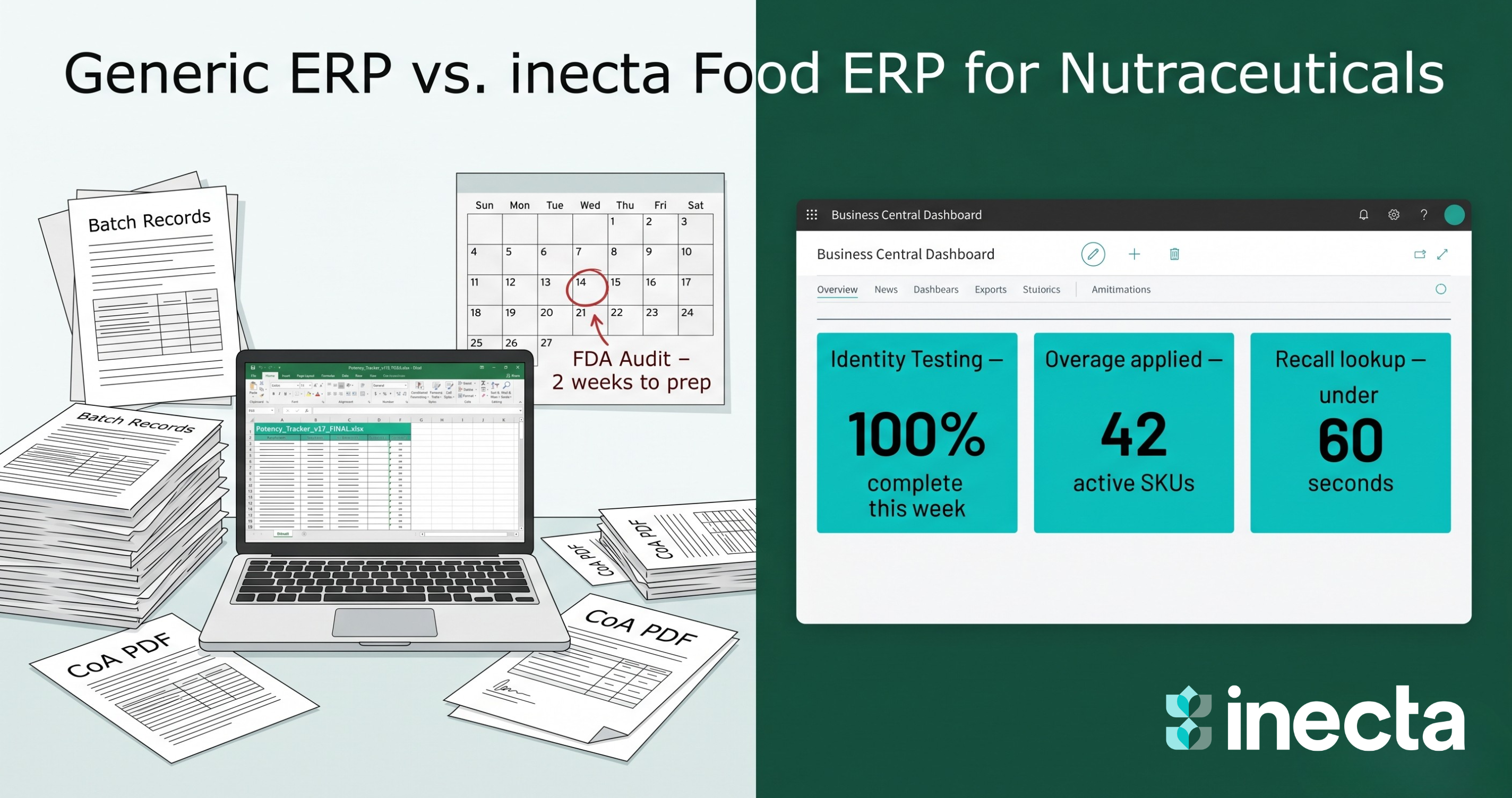
Most ERP systems were built for distribution and discrete or process manufacturing in the abstract. They handle inventory, purchase orders, production scheduling, and accounting well but supplement manufacturing has requirements those generic platforms simply do not include out of the box:
- Potency variability in natural ingredients. A turmeric extract lot tests at 93% curcuminoids one week and 96% the next. The ERP needs to record the actual assay value for the incoming lot and dynamically recalculate batch quantities so the finished product hits its label claim.
- Overage management. Active ingredients degrade over shelf life. Manufacturers formulate with a calculated surplus so the label claim still holds at expiration. The system needs to store overage percentages per active ingredient, apply them to every batch, and verify the math at release.
- Identity testing documentation at receiving. Part 111.75 requires this for every dietary ingredient. The ERP needs to capture the test method, limits, results, and QC approval and link them to the specific lot in inventory.
- Automated Supplement Facts and COA generation tied to formulation and test data, with version control on labels and substantiation files for structure/function claims.
- Hold-and-release workflows that quarantine non-conforming material at every checkpoint without manual intervention.
Bolting these capabilities onto a generic ERP through customization is expensive, fragile, and creates technical debt that breaks every time the underlying platform updates.
How inecta Food ERP Is Built For 21 CFR Part 111
inecta Food ERP is a purpose-built food and beverage ERP with a dedicated nutraceutical product line, running on Microsoft Dynamics 365 Business Central. Part 111 compliance is not a module you buy separately or a customization you commission it is part of how the system was designed.
Dedicated 21 CFR Part 111 module. Captures every required cGMP record in one place: master manufacturing records, batch production records, component examination and identity testing at receiving, in-process testing, finished product testing, and product release. When auditors arrive, every record is one query away not days of pulling from spreadsheets and email.
Formula and recipe management with active-ingredient tracking. Manage multi-layer formulations with unit-of-measure conversions across mg, mcg, IU, and CFU. Reformulate once and the system updates yields, costs, and label claims everywhere.
Potency and overage tracking as a named feature. Store overage percentages per active ingredient based on stability data, incorporate them into every batch calculation, and verify each finished lot meets label claims at manufacture and through end of shelf life. This is the substantiation backbone for DSHEA label claims.
Full lot traceability. Every raw material lot, intermediate blend, and finished good is tracked forward and backward across the supply chain. In a recall scenario, identify every affected lot, customer, and shipment in minutes meeting both DSHEA and FSMA requirements.
Quality control checkpoints with automated COA generation. Hold-and-release workflows quarantine non-conforming material at receiving, in-process, and finished product stages. Certificates of Analysis pull directly from in-system test data no manual transcription, no version drift between QA and shipping.
Contract manufacturing and private label support. Manage customer-specific formulas, separate quality specifications per brand, and track cost and margin at the product, customer, and order level all with the same compliance backbone.
What Audit-Ready Actually Looks Like
The difference between a stressful FDA inspection and a routine one usually comes down to how long it takes to produce a record. With Part 111 compliance built into the ERP, "audit-ready" stops being a quarterly fire drill and becomes the default state of the business. Every batch record ties to its master record. Every identity test ties to its component lot. Every COA ties to its quality data. Every shipment ties to its traceable lot history. Nothing is reconstructed after the fact, because nothing is ever scattered in the first place.
That is what 21 CFR Part 111 actually asks of a manufacturer not heroic documentation effort, but a system of record that makes compliance the byproduct of daily operations.
Ready To See It In Action?
inecta Food ERP is trusted by nutraceutical manufacturers including Timeline Nutrition and Barrington Nutritionals, with a 99% customer retention rate and 25+ years of food and beverage industry experience. If your supplement operation is still bridging Part 111 with spreadsheets and email, book a discovery call and we will show you what built-in compliance looks like for your specific products and production model.